These substances belong to a broad category of per- and polyfluorinated alkyl substances (PFAS). They can be used as surfactants, exhibiting excellent hydrophobic and oleophobic properties and are used in a variety of industrial applications. However, these substances can pose a threat to humans and the environment, leading international market regulators to gradually regulate them.

These substances belong to a broad class of per- and polyfluorinated alkyl substances (PFAS). They can be used as surfactants, exhibiting excellent hydrophobic and oleophobic properties, and are used in a variety of industrial applications. However, these substances pose health and environmental risks, leading international market regulators to gradually regulate them. Perfluorocarboxylic acids (PFCAs), their salts, and related compounds are compounds with the general formula CnF(2n+1)-COOH, CnF(2n+1)-COOX', or CnF(2n+1)-X, where X' = any group and X = any group other than F, Cl, and Br. Their chemical structure primarily consists of carbon atoms bonded to fluorine atoms. The strong carbon-fluorine bond contributes to the extreme stability of these substances. This type of substance belongs to a large class of per- and polyfluorinated alkylated substances (PFAS). It can be used as a surfactant and has good hydrophobic and oleophobic properties. It is used in a variety of industrial applications. However, these substances are harmful to humans and the environment. Therefore, the competent authorities of various markets around the world have gradually included them in the regulation.
Introduction to the Perfluorocarboxylic Acid (PFCA) Family
When discussing PFCA, it's inevitable to mention its related parent, PFAS. The PFAS family of chemicals is vast, comprising over 4,700 fluorine-based compounds. PFCA and its relatives, PFOS and PFOA, are members of this family. A simple chart will help you understand their relationship.
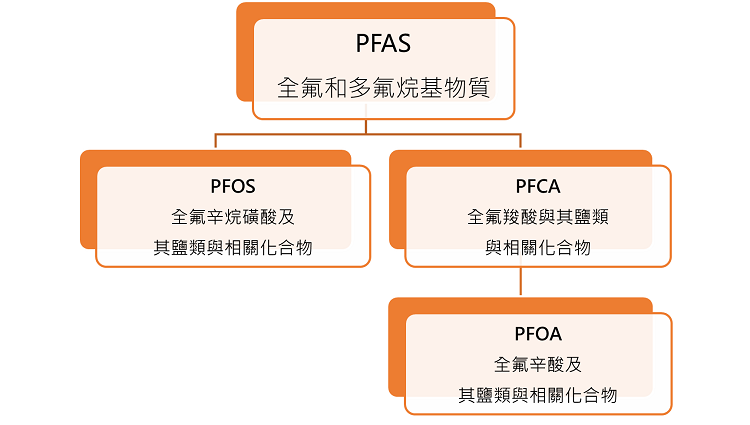
Why is the world starting to pay attention to perfluorocarboxylic acids (PFCAs), their salts, and related compounds?
♦Perfluorocarboxylic acids (PFCAs), their salts and related compounds
Perfluorocarboxylic acids (PFCAs), their salts, and related compounds are widely used in non-stick cookware production as surfactants, wire insulation, surgical implants, pipe sealing tapes, photolithography and etching processes in semiconductor manufacturing, firefighting foams, and waterproof, anti-fouling, and non-stick coatings due to their stability and hydrophobic and oleophobic properties. They are also widely used in the production of fluoropolymers and fluororubbers. The international attention garnered by PFCA compounds cannot be overstated without mentioning polytetrafluoroethylene (PTFE), which is synthesized using PFCAs as a raw material. PTFE is resistant to acids and alkalis, high temperatures (compared to other polymers), and various organic solvents. Its excellent properties have led to its use in a variety of industrial and commercial applications, particularly in waterproof, anti-fouling, and non-stick coatings.
♦The world-famous perfluorooctanoic acid (PFOA) pollution incident:
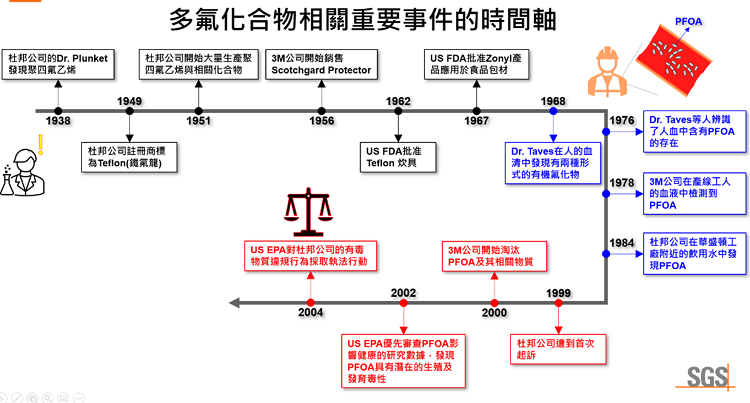
Polytetrafluoroethylene (PTFE) was discovered in 1938 by Dr. Plunket of DuPont, who accidentally synthesized it while attempting to create a new chlorofluorocarbon. The PTFE manufacturing process uses perfluorooctanoic acid (PFOA), a perfluorocarboxylic acid (PFCA) compound, as a surfactant. After the process is complete, PFOA remains in the synthesized PTFE and in the reaction waste. These PFOA residues have led to a century-long environmental pollution incident. After years of research, DuPont registered the trademark for polytetrafluoroethylene (PTFE) as Teflon in 1949 and began mass production at its Washington, D.C., plant in 1951. 3M supplied the raw material perfluorooctanoic acid (PFOA) required for the synthesis process. Since then, polytetrafluoroethylene (PTFE) has been widely used in various fields, generating staggering profits for companies. However, the large-scale production and use of PTFE has also brought hidden problems to the surface.
In 1968, Dr. Taves first discovered two forms of organic fluoride in human serum. In 1976, Dr. Taves and others identified the presence of perfluorooctanoic acid (PFOA) in human blood. This discovery attracted the attention of 3M and DuPont, who began private research on the effects of PFOA on animals, humans, and the environment. In 1978, 3M detected PFOA in the blood of production line workers. In 1984, DuPont discovered PFOA in drinking water near its Washington, D.C., plant. Between 1980 and 1987, 3M and DuPont continued to study PFOA. Although PFOA was known to accumulate in the natural environment and cause adverse effects on organisms, the huge profits led to the continued large-scale production and use of polytetrafluoroethylene (PTFE).
It wasn't until 1999, when DuPont was sued for long-term burial of PFOA-containing reaction waste in a landfill, resulting in the deaths of cattle on nearby farms, that the issue began to attract government attention. In 2000, 3M announced its intention to phase out PFOA and related substances based on responsible environmental stewardship principles. In 2002, the U.S. Environmental Protection Agency (EPA) prioritized its review of research data on the health effects of PFOA and found it to have potential reproductive and developmental toxicity. In 2004, the EPA began taking enforcement action against DuPont for toxic substance violations.
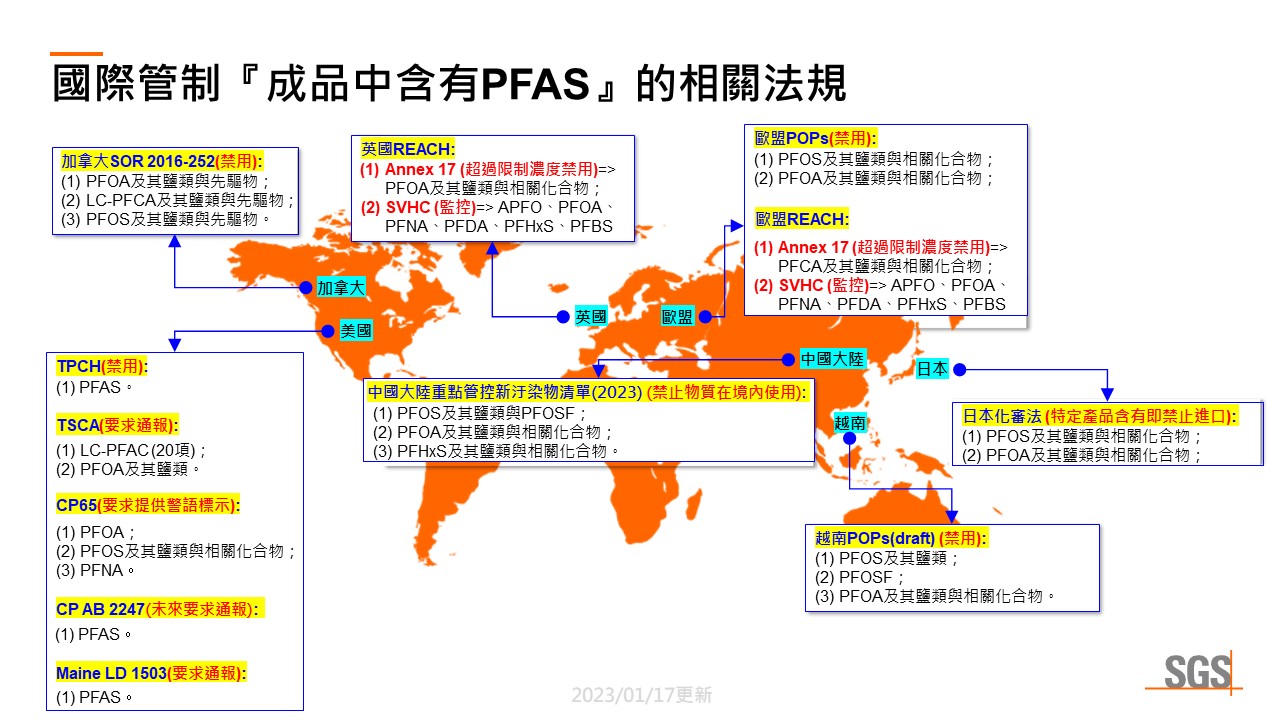
Common international regulations governing perfluorocarboxylic acids (PFCAs), their salts, and related compounds:
The world-renowned Persistent Organic Pollutants (POPs) pollution incident has garnered international attention on perfluorocarboxylic acids (PFCAs), their salts, and related compounds. Regional authorities have gradually incorporated them into chemical regulations to protect citizens and the environment. Given the varying regulations of various market authorities, SGS has compiled the following common international regulations governing the presence of perfluorocarboxylic acids (PFCAs), their salts, and related compounds in finished products:
EU:
- EU REACH (Regulation (EC) No 1907/2006)
- Annex 17 of the Hazardous Substances List - Controlled Substances, Mixtures, and Finished Products. The following substances are prohibited from sale if their concentration exceeds the limit:
- Total concentration of C9-C14 perfluorocarboxylic acids (PFCA) and their salts: 25 ppb
- Total concentration of C9-C14 perfluorocarboxylic acids (PFCA) and their salts: 25 ppb
- Substances of Very High Concern (SVHC) - Controlled concentrations of the following substances in finished products. Exceeding these concentrations will require follow-up actions according to regulations.(詳細內容):
- Perfluorooctanoic acid (PFOA), ammonium perfluorooctanoate (APFO), perfluorononanoic acid (PFNA), and nonadecafluorodecanoic acid (PFDA), and their sodium and ammonium salts: 1000 ppm
- EU POPs (Regulation (EC) No 2019/1021)
- Annex I-管制物質、混合物與成品中以下物質濃度,超過限制濃度即禁止販賣:
- Total concentration of perfluorooctanoic acid (PFOA) and its salts: 25 ppb
- 全氟辛酸(PFOA)相關化合物總濃度: 1 ppm
U.K:
- UK REACH (The REACH etc. (Amendment etc.) (EU Exit) Regulations 2020 No. 1577)
- Annex 17 of the Hazardous Substances List - Controlled Substances, Mixtures, and Finished Products. The following substances are prohibited from sale if their concentration exceeds the limit:
- Total concentration of perfluorooctanoic acid (PFOA) and its salts: 25 ppb
- Total concentration of perfluorooctanoic acid (PFOA) related compounds: 1 ppm
- Substances of Very High Concern (SVHC) - Controlled concentrations of the following substances in finished products. Exceeding these concentrations will require follow-up actions according to regulations.
- Perfluorooctanoic acid (PFOA), ammonium perfluorooctanoate (APFO), perfluorononanoic acid (PFNA), and nonadecafluorodecanoic acid (PFDA), and their sodium and ammonium salts: 1000 ppm
Canada:
- SOR 2016-252
- The following substances are prohibited from being contained in controlled substances, mixtures and finished products:
- Perfluorooctanoic acid (PFOA), its salts and precursors
- C9-C20 perfluorocarboxylic acids (PFCAs) and their salts and precursors
USA:
- Toxic Substances Control Act (TSCA)
- Surface coatings on regulated finished products must notify the U.S. Environmental Protection Agency (EPA) at least 90 days before importing or processing any of the following substances:
- Long-chain perfluoroalkyl carboxylates (LC-PFAC)
- Perfluorooctanoic acid (PFOA) and its salts
- Model Toxics in Packaging Legislation (TPCH)
- The following substances are prohibited from being contained in regulated packaging materials:
- Perfluoroalkyl and polyfluoroalkyl substances (PFAS)
- California Proposition 65 (CP65):
- Control exposure to hazardous substances in consumer products, including inhalation, ingestion, skin contact or other exposure routes, requires warning labels to be provided when exceeding the safety protection value:
Japan:
- Chemical Substances Control Law (CSCL):
- Specific products such as water- and oil-resistant paper, textiles and clothing, detergents, anti-reflective agents for semiconductor manufacturing, paints, varnishes, adhesives, fire extinguishers, etc. will be prohibited from import if they contain the following substances:
- Perfluorooctanoic acid (PFOA) and its salts
Vietnam(Draft, expected to take effect on January 1, 2023):
- National Technical Regulation (draft):
- 管制物質、混合物與成品,以下物質超過限制濃度即禁止販賣:
Internationally, in addition to regulating perfluorinated carboxylic acids (PFCAs), their salts, and related compounds, other related compounds in perfluoroalkyl substances (PFAS), such as perfluorooctane sulfonates (PFOS) and perfluorohexane sulfonate (PFHxS), are also regulated in national and regional regulations.